Nastic Acuators
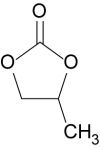
Propylene Carbonate as a Pumping Fluid
On this page:
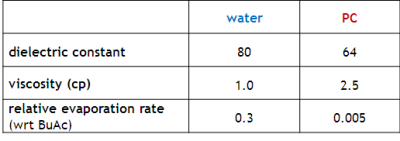
Deepa Sritharan, with the assistance of undergraduates Simpson Chen, Prabhath Aluthgama, and Bilal Naved, showed that without added salt, PC is stable to thousands of volts. The figure below compares electroactive steel electrodes in water and PC under 3000 V. (This is not the case for PC with salts.) Electrokinetic Pumping with PC Flow rates are comparable to those of water, while response speeds are a bit faster and pressures are higher.
For further information, see: 1. D. Sritharan, A. S. Chen, P. Aluthgama, B. Naved, and E. Smela, "Bubble-free electrokinetic flow with propylene carbonate," Electrophoresis (in press) (2015). Deepa Sritharan is now working on paper-based EOF devices, including afabrication method based on lamination.
|
|
|
©2013